Built on real regulatory workflows
Not generic templates. The platform mirrors how teams actually prepare medical device documentation — step-by-step, with structured tasks, organised folders, and clear "what to include" guidance.
Getting your medical device CE marked can feel complex. We break the process down into clear, simple steps — from a free MDR check to a full regulatory workflow, with built-in AI support.

Backed by

Breaking down regulatory compliance into bite-sized pieces.
Tell us what you're building and who it's for. We turn your input into a clear description that fits regulatory expectations.
We show you if MDR applies, what risk class you fall into, and the route you'll need to take.
Follow a structured plan to create your documentation step by step. Instead of starting from scratch, you get guidance, templates, checklists, and a clear sense of direction.
Start free. Upgrade when you're ready.
Trial
Free
no credit card required
Pro
€299
per month · excl. VAT
Pro Annual
€2,990
per year · Save 2 months · excl. VAT
Shaped by regulatory specialists. Grounded in official EU guidance.
Not generic templates. The platform mirrors how teams actually prepare medical device documentation — step-by-step, with structured tasks, organised folders, and clear "what to include" guidance.
From day one, you work in a reviewable structure. Track progress, maintain consistency across documents, and reduce gaps that slow down internal reviews.
Know what's required — and why. Every step helps you understand the rationale behind decisions, so you can align stakeholders and defend choices with confidence.
Plan, draft, and maintain your MDR documentation in one guided workflow.
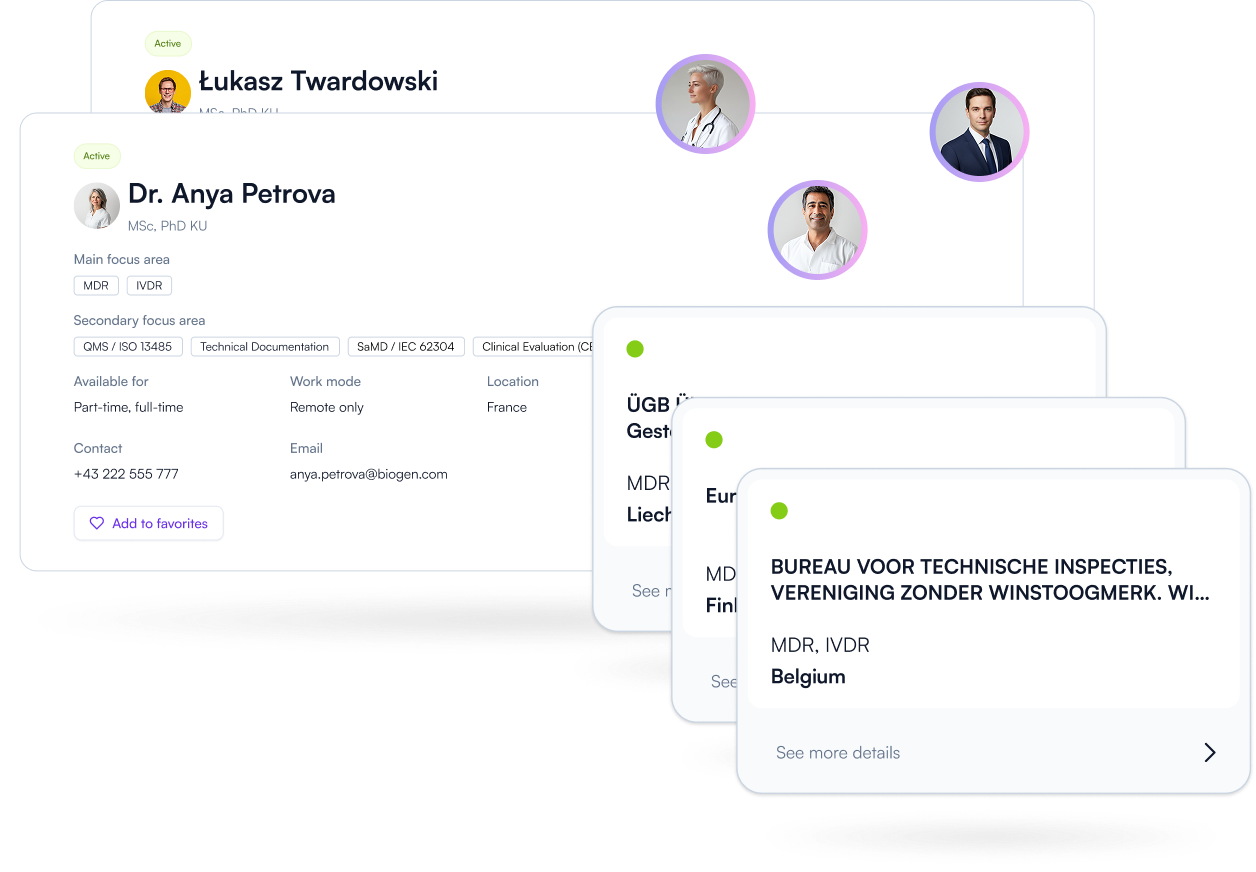
Find the right experts faster. Compare specialization, scope, and collaboration models — connect when your project needs regulatory support. Less searching, better fit, faster progress.
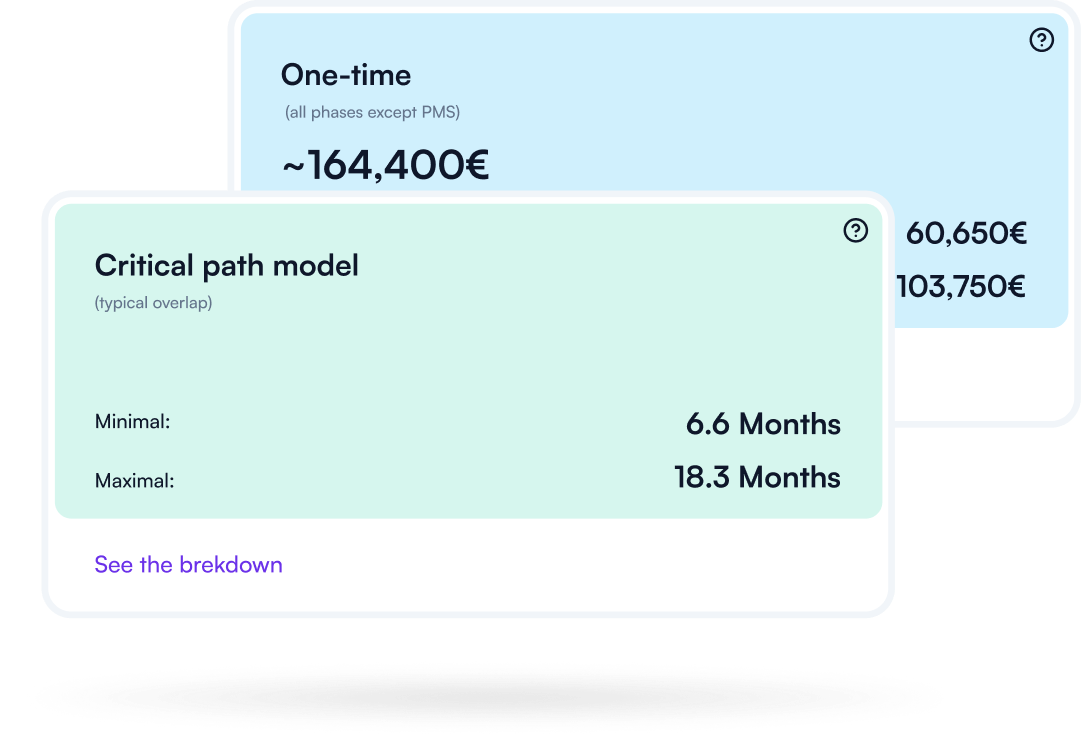
Understand what drives effort and cost across your compliance journey — so you can plan realistically, prioritize, and avoid late surprises. Make timelines and budgets defensible.
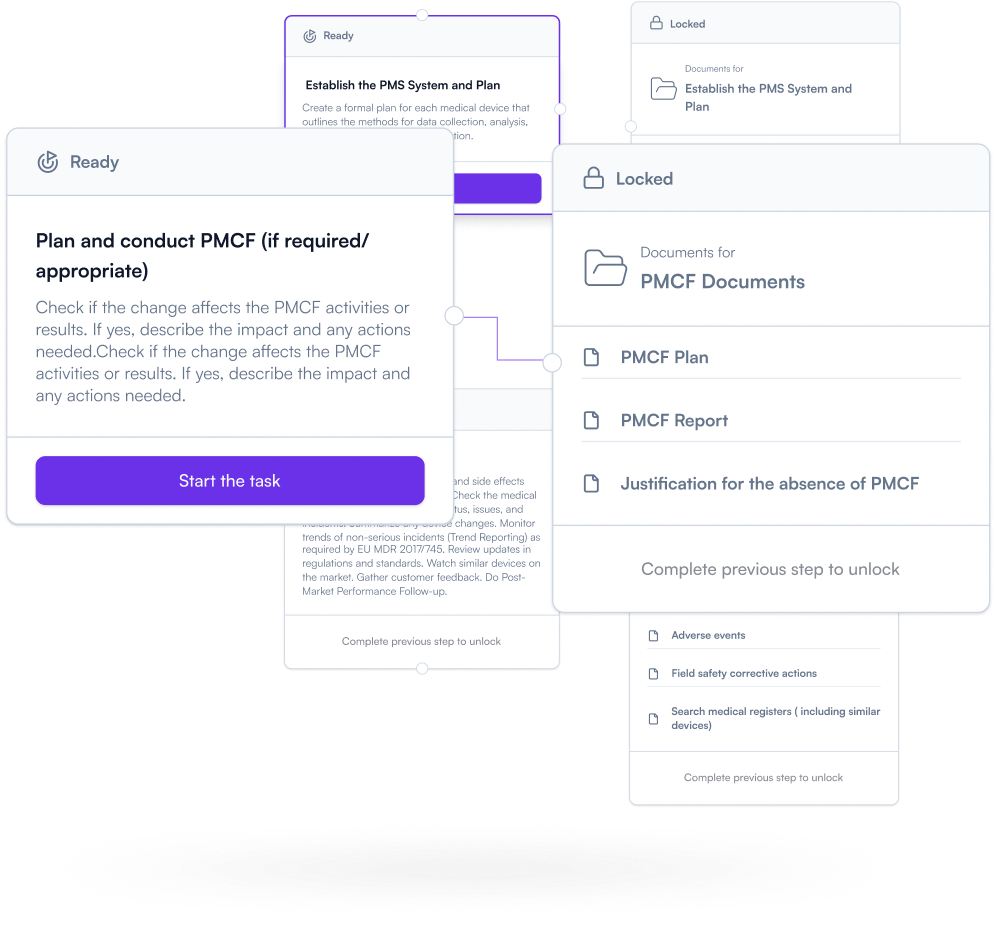
Don’t treat PMS (Post-market surveillance) as an afterthought. Plan your post-market activities early and keep documentation aligned as real-world feedback and changes come in. Keep your PMS evidence audit-ready — continuously.
Insights on health tech regulation, MDR compliance, and medical device pathways.

Why starting the regulatory conversation early is the smartest decision a health tech founder can make. Health tech founders think about risk constantly. Market risk, technical risk, funding risk. There is one that almost never makes the list. And it is the one most likely to stop you. It builds quietly. A vague product description. Pilots run without regulatory structure. A quality system nobody actually uses. None of it feels like a crisis at the time. Together, it becomes a problem that c

The Notified Body has not gone silent because your file is bad. It may have gone silent because no one told you what to expect, and no one inside is obligated to update you. That is not a bug in the process. It is the process. Most founders manage this quietly at first. A missed milestone gets reframed as a minor slip. A verbal estimate from the NB gets passed to the board as a commitment. The consultant adds a line item. Each thing seems handleable in isolation. Together, it becomes a problem

Here is the rewritten blog post: Most founders think clinical evaluation is a document. It is not. It is a process, and Annex XIV of the MDR defines exactly what that process requires. The founders who treat it as paperwork tend to discover the difference at the wrong moment. You write your evidence strategy around a competitor's device. You sketch a plan to handle PMCF after launch. You assume equivalence will hold. Each of those decisions seems reasonable in isolation. Together, they become
Clear answers for teams building regulated products.
Book a demo with one of our team members
Whether you're just getting started or already building, get clarity on your regulatory path and access the full workflow with Health Tech Pathways.